AuscultaNet Joins SITEM MedTech Booster
Selected for Switzerland's leading MedTech acceleration program, providing regulatory mentorship, clinical infrastructure, and access to the Swiss hospital network.
AuscultaNet transforms standard stethoscope recordings into actionable cardiac insights. Offline AI detects and classifies heart murmurs — including timing classification no other system provides.
Today, 56% of significant heart valve disease goes undetected in primary care. AuscultaNet changes that.
of significant valve disease goes undetected by primary care physicians
Every diastolic murmur requires immediate cardiology referral, yet they are among the most frequently missed findings.
Without timing classification, physicians cannot distinguish between innocent flow murmurs and those requiring intervention.
Studies show general practitioners detect fewer than half of clinically significant murmurs during routine auscultation.
Standard 4-site cardiac auscultation with any digital stethoscope. Under 5 seconds per site.
On-device AI processes the recording instantly. No cloud, no internet, no patient data leaves the device.
Receive murmur detection, timing classification, grade estimation, and a prioritised clinical recommendation.
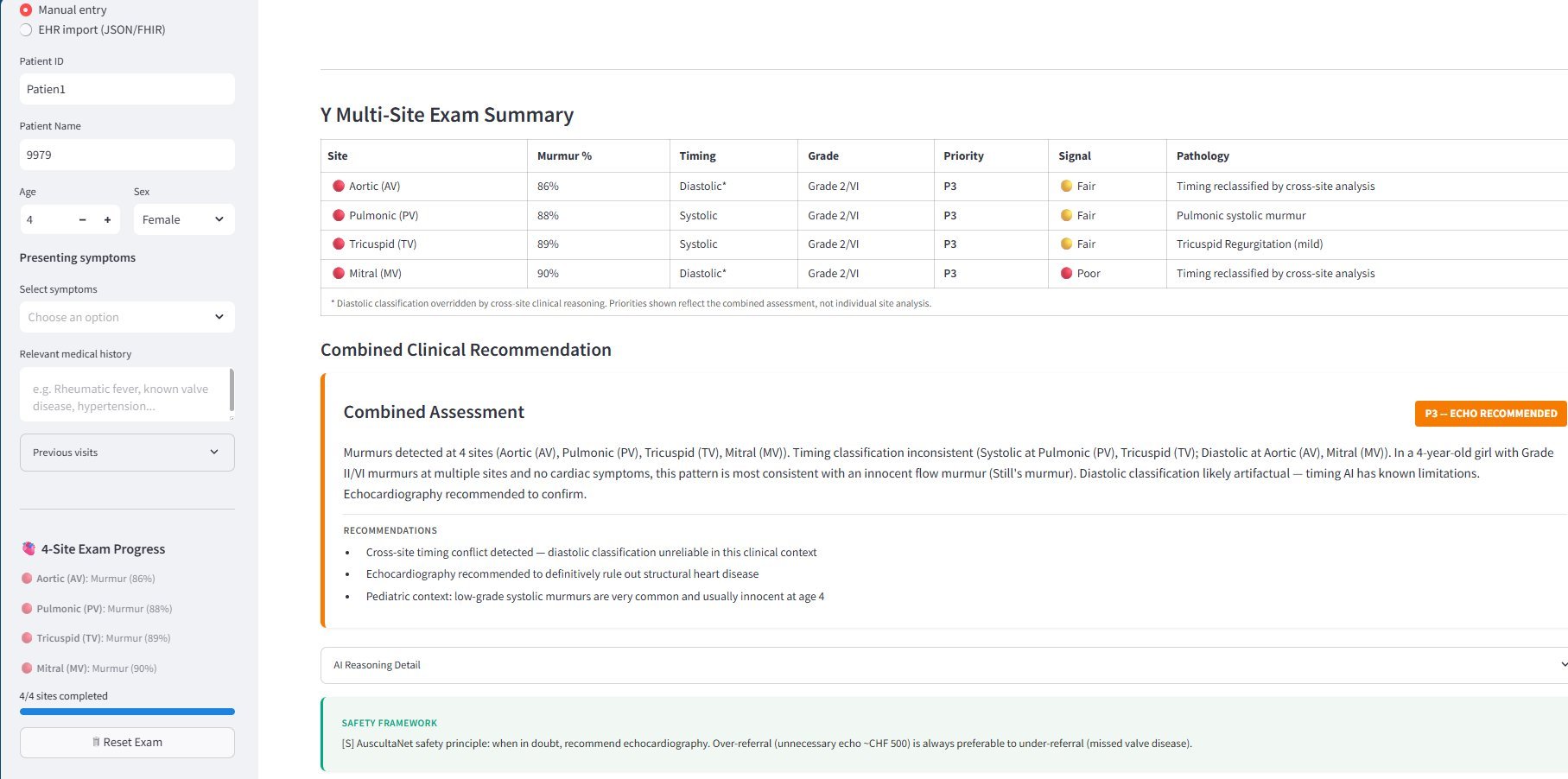
AuscultaNet clinical interface — 4-site examination with combined clinical reasoning
Classifies murmurs as systolic, diastolic, or continuous — the clinical distinction that determines urgency of referral.
All AI processing on-device. Complete data sovereignty. No infrastructure dependencies.
Validated on public, reproducible datasets. Performance independently verifiable.
Structured examination across Aortic, Pulmonic, Tricuspid, and Mitral sites with cross-site clinical reasoning.
Built-in escalation protocols. When in doubt, recommend echocardiography.
FHIR R4 and HL7 compatible. Structured export for Swiss EPD and international EHR systems.
Multi-task clinical model with 7 simultaneous outputs. Tested on standardised, publicly available data with proper patient-level splits and bootstrap confidence intervals.
95% CI: 0.893 – 0.939 • Bootstrap n=1000
AuscultaNet is developed in Switzerland, supported by leading institutions for MedTech innovation, deep tech, and startup growth.
A focused team combining AI engineering, regulatory strategy, and clinical cardiology.

13+ years in safety-critical systems (automotive & MedTech). Experience with MDR and FDA 510(k) submissions. Leads regulatory and clinical strategy.
LinkedIn
Data & AI Engineer, Embedded System Architect, Azure-certified. Leads AI integration and device architecture.
LinkedIn
20+ years in medical device regulatory affairs. Former Ypsomed AG, leading FDA 510(k) and CE Mark submissions. Deep expertise in ISO 13485 QMS, EU MDR, and US market access.
LinkedIn
Consultant cardiologist. Leads pilot study planning in Greece and Cyprus. Signed LOI.
LinkedInSeed round open to fund clinical validation and regulatory clearance. Join leading MedTech-focused investors in closing the cardiac diagnostic gap.
Join our prospective multi-site validation study. We are recruiting partner institutions for a clinical trial comparing AuscultaNet to echocardiography as gold standard.
Join us in closing the diagnostic gap in primary care.
We would love to hear from you.
We are raising a seed round to fund clinical validation and regulatory preparation.
Join our prospective validation studies. Co-authorship opportunities available.